Karl.Fisher using Karl Fischer reagent for determination of moisture content-method, quantitatively, the reagents to selectively react with water.
Karl Fischer reagent composition is iodine, sulfur dioxide, base and solvent, such as alcohol.
I2+SO2+3Base+ROH+H2O = 2Base·HI+Base·HSO4R……(1)
As described below, this method can be used for volumetric and coulometric titration system.
Coulometric titration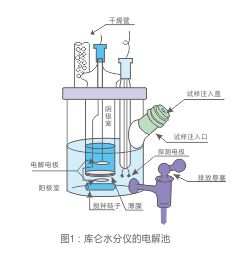
Coulometric titration.Sample addition in the electrolyte, its main ingredient is iodide ions, sulphur dioxide, salt, as well as solvents (such as alcohol).Such as equation (2) shows, produced by electrolytic oxidation of iodine, and Karl Fischer reagent react directly.
2I- - 2e = I2 ……(2)
According to Faraday's law, is proportional to the power of iodine.This means that moisture can directly from the electrolytic oxidation of the Coulomb number you want it.
1mg的水=10.71库仑
As shown in Figure 1, you need two types of coulometric reagent: an electrolyte is anode, located in cell anode Chamber, and a cathode electrolyte, placed on the electrolytic cell, the cathode Chamber.Coulometric reagent does not need to be assessed.Another benefit is that you can use again and again.
Capacity titration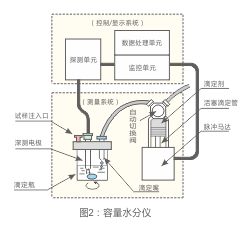
In a flask in a suitable solvent dehydration of the specimen.With the titrant transferred all the moisture from the solvent.Then add specimen.Titration with titrant, titer (mgH2O/mL) is determined in advance.The moisture content is determined by titration of the sample volume (mL) is determined.Using a constant polarization voltage detecting end points.Figure 2 shows a typical marketing Automatic volumetric titration of the various components of the system.
